Why Coatings Are Not a Good Solution for Rotating Equipment
-
simsite
-
July 26, 2023
To understand why coatings are not a good solution for rotating equipment, we must first explore the primary reason for coating any metallic part. The reason is to prevent corrosion attack. However, there are many different types of corrosion and we should look at how each type of corrosion can affect coatings on metallic surfaces to determine if this process is worthwhile for the required service life.
(a) Uniform Corrosion. – Corrosion that occurs uniformly over the piece and results in rust and metal loss over the entire metallic surface.
(b) Galvanic Corrosion. – Corrosion that occurs as a result of two or more dissimilar metals that are electrically coupled together by the same medium (fluid).
When a galvanic couple forms, one of the metals in the couple becomes the anode and corrodes faster than it would all by itself, while the other becomes the cathode and corrodes slower than it would alone. The metal with the lower nobility becomes sacrificial.
(c) Crevice Corrosion. – Crevice corrosion is a form of localized corrosion that occurs in crevices where the surface environment differs from the surrounding surface area.
The different surfaces result in corrosion because of differences in concentration of oxygen, pH, and ferric ions. If there is an oxygen concentration difference, corrosion will accelerate at crevices where there is less oxygen than in the environment surrounding the crevice. Crevices are formed when two surfaces are in close proximity to one another, such as when two metal surfaces are against one another, even when a gasket is against the surface.
Crevice corrosion can occur under grease, slime, and coatings on any metallic surface.
(d) Pitting corrosion. – Pitting Corrosion is a localized form of corrosion which causes “cavities” or “holes” to be produced in the material. Pitting is considered to be more dangerous than uniform corrosion damage, because it is more difficult to detect, predict and protect against. Pitting is caused by the following:
a. Localized chemical, or mechanical damage to the protective oxide film caused by low dissolved oxygen concentrations which breakdown the oxide, and/or high concentrations of chloride as in seawater.
b. Localized damage to the metallic product, or poor applications of protective coatings.
c. The presence of non-uniformities in the metal structure of the component.
(e) Cavitation corrosion. – The metal loss caused by the formation and collapse of vapor bubbles in a liquid near a metal surface. Cavitation occurs when a fluid’s operational pressure drops below the vapor pressure of the liquid it is pumping causing gas pockets and bubbles to form and collapse.
This form of corrosion will eat out the volutes and impellers blades of centrifugal pumps in a very short period of time.
Coatings are rendered useless against this form of corrosion.
(f) Erosion-corrosion. – The accelerated metal loss from an initial corrosion mechanism associated with high-velocity flows and abrasion. Erosion-corrosion is caused by the relative movement between a corrosive fluid and a metal surface. This process leads to the formation of grooves, gullies, valleys, rounded ridges, wavy surfaces, holes, etc., and exhibits a directional flow pattern. (Comet tails, horseshoe marks, etc.)
In two-phase liquids (containing suspended solid particles, solids, or gas bubbles), the impact of the particles damages, or even eliminates the protective layers, passive films, or protective coatings causing the local corrosion rate to be markedly accelerated. This phenomenon is called abrasion-corrosion.
(g) Dealloying. – Dealloying is an electrochemical reaction in which one element constituting an alloy is selectively separated and removed from the alloy, causing deterioration of its essential properties. Dealloying is an unusual type of corrosion, occurring mainly in certain alloy metals such as copper alloys as well as in gray cast iron. When the dealloying takes place, the alloy metal loses its reactive element and retains the remaining alloy in a porous state.
For example, for a brass alloy, the reactive component, zinc, becomes the anode and is selectively removed due to galvanic corrosion, while copper remains in a spongy, porous surface. This process is called Dezincification. – The selective dissolution of zinc from brass alloys. It is recognized by a color change (e.g., from its original yellow brass color to a distinctly red, coppery appearance). The properties, such as mechanical strength, of the remaining material are significantly reduced and can fail during normal operation.
In the case of cast iron, the main constituents are iron and graphite, and graphite becomes the anode to be removed by galvanic action. This process is called Degraphitization.-The selective dissolution of iron from cast iron, usually gray cast irons. It normally proceeds uniformly inward from the surface, leaving a porous matrix alloy that is composed mostly of carbon. Degraphitization can be recognized by a change from an original silver-gray color to a dark gray. The affected metal can be easily cut or pierced.
Coating Effectiveness — The effectiveness of a coating depends on many factors – matching the coating to the intended service exposure, the type of preparation for the coating, the type of coating, the type of corrosion involved, the velocities the part will be exposed to, the type of fluid, and the amount of abrasive particles.
When you look at most coatings under magnification, you will see tiny cracks, pits, and crevices. These defects quickly allow for concentrated corrosion attack compromising the coating. The quality of the coating, preparation, and application become critical to the life of any coating.
Pump Applications – Service Exposure. – While coatings might work for short periods of time, they may not be the best option for the long term protection of your Impellers, Pump Casings, and other Liquid End Parts. As you can see from the above corrosion explanations, coatings are short term solutions, which may not work well for certain types of corrosive situations such as cavitation corrosion. Today, there are better solutions against corrosion and cavitation on the market that NEVER Corrode, such as structural composites like SIMSITE® Impellers & Rings, Pump Casings, and other Liquid End Parts. These parts are not affected by corrosion of any type.
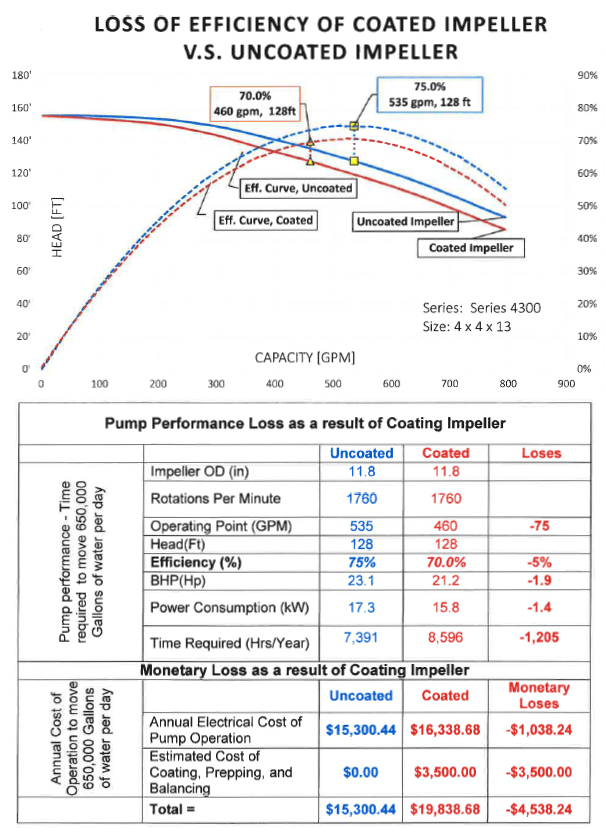
Reduction in Efficiency
As you can see in the above Charts and Graph, Coating the Metallic Impeller caused a narrowing of the impeller vane channels, which reduced the Operating Point to be reduced from 535 GPM to 460 GPM.
The coating also caused the operating point to be moved back on the Head-Capacity Performance Curve (HQ curve) farther away from the Best Efficiency Point (BEP). As a result, Pump Efficiency was also reduced by 5% from 74.8% to 70.0%. In order to pump the same amount of fluid, the pump with the coated impeller must work an additional 1,205 hours per year. The result is a total monetary loss of $4,538.
In addition to the loss of efficiency, the coating caused the operating point to be moved back on the HQ Performance Curve away from the Best Efficiency Point (BEP) which increases radial and actual movement. This increase in radial and actual movement of the impeller increases vibration and cavitation in the pump and accelerates wear on the motor bearings, mechanical seal faces and rings.
Velocity of the Liquid being Pumped – The velocities of the liquid play a critical part in determining whether or not to use a protective coating. It in high velocity areas, coatings become only temporary short terms fixes. For high velocity pumping conditions, or for higher speed rotating equipment coatings are usually not a good solution, because they will quickly be removed from the metallic part. Whereas, for stationary equipment, or for low velocity, low RPM rotating equipment coatings may be an effective solution. The effectiveness of the coating depends upon the preparation prior to coating, the application of the coating, and the quality of the coating.
Coatings vs Structural Composites
At Sims, we receive many questions about coatings pertaining to pumps and rotating equipment. Do coatings really work? Are some coatings better than other coatings? And how efficient is a coating for an impeller? Does it protect the impeller?

To answer the above questions, it is necessary to consider the application, the type of service, the part, and the velocities that the part will experience. Coatings normally work well for stationary items such as pump casings that will not see high turbulence, velocities, or erosion from abrasive particles immersed in the fluid. For pump casings, it does no good to coat the casing if the coating will be removed around the area of the “Cut Water,” which experiences the highest velocities and turbulence. Wearing the “cut water” substantially reduces efficiency and performance.
While coatings might work well for stationary items, they are not usually a viable option for the long term protection of your Impellers, and other “rotating” pump parts. Rotating equipment such as impellers experience many more stresses and movements in different directions. The movement combined with high turbulence and velocities make protective “coatings” not the best choice for Impellers and rings.
The pictures (above) show how poorly these two different types of ceramic/epoxy coatings have protected these Metallic Impellers. These ceramic coatings cracked and chipped; leaving the Metallic Impellers exposed to corrosion and degradation to the point where holes can be seen through the impeller vanes on both impellers. The coatings fail where the high liquid velocities and vortexes are presented. The higher the velocities of the fluid the faster the coatings fail, or are removed from the metallic impellers.
As a result of the failure of the coatings to adhere to the impellers, the coatings failed in their job to prevent corrosion and damage to the Metallic Impellers, and the coatings failed in their attempt to extend the life of the impellers. The type of damage done to these impellers renders them highly inefficient and leaves the impellers unusable. Coatings are not the best solutions to protect metal when dealing with high velocities, vortexes, or cavitation.
Better Solutions for Pump Applications. Structural Composite Impellers, pumps, and pump parts are not affected by corrosion, and will never corrode due to non-corrosive nature of the Composite. The properties of structural composites enable them to last a very long time in salt water without degradation of material properties. High liquid velocities and vortexes are not affecting the structural composite impellers, because they are engineered and machined to eliminate problems. Structural composites like Simsite® can last many times the life of coated metallic impellers. Most metals cannot survive the effects of corrosion once the liquid gets under the coating. Adhesion, pitting and stress cracking are constant problems when applying coatings onto metallic impellers.
With the possible difficulties applying coatings and with a possibility of a very limited lifetime, coatings may not be the best option when it comes to choosing your impellers. Structural Composites do not need coatings and last much longer than coated metallic impellers in corrosive applications. Below you can see a Simsite® Structural Composite impeller that can last easily last 15 to 20 years in sea water service.

Simsite® Impellers are machined from solid blocks of Simsite® structural composite on 5-8 axis machining centers and therefore are energy efficient, light weight, have perfect balance, and inherent non-corrosive properties.
By John A. Kozel, the President of the SIMS Pump Valve Company, Inc.
Resources
–Toward Polymeric and Polymer Composites Impeller Fabrication
–Wastewater Technology Fact Sheet: In-Plant Pump Stations
–Wastewater Technology Fact Sheet: In-Plant Pump Stations
–Manufacturing of Closed Impeller for Mechanically Pump ..
–Corrosion and failure of service water pump impeller snap rings
–Right Pump Impeller Design Changes to Improve
–Trim or Replace Impellers on Oversized Pumps
–Design of pump impellers using digital computer
–Submersible Multistage Centrifugal Pump for Versatile Testing

















